
Time:2017-11-23| Author:admin
From the following aspects, this paper explains the contrast of the BDS-5000 hydrogen peroxide steam sterilization equipment produced by our company and other similar types of main hydrogen peroxide sterilizers.
Sterilization process:
First, dehumidification
There are some other brands of hydrogen peroxide sterilizers, mainly imitating the US STERIS company products, and its principles are "dry" sterilization, which requires dehumidification operation in the pre-sterilization of room or clipping. To achieve the relative humidity of the set, it is generally 40% and below; our BDS-5000 is "wet" sterilization theory, that is, the sterilized room or hatter is not required in the early stage of the sterilization. operate. This process, there is a point you need to pay attention:
1, time consuming:
Compared with the predecessor of the BDS-5000 without the need for dehumidification, the process of dehumidification has greatly increased the time of sterilization, and it has greatly increased the time of downtime, which is extremely unfavorable in the case of the production period;
2, energy consumption:
Dead wet operation consumes more electrical energy
3, the cost is higher:
Diffuring functions require additional equipment, increase the cost of purchase, and the cost of future maintenance;
4, system complexity:
Our technology does not need to dehumidify, normal environmental conditions, and under the premise of ensuring more excellent killing, dehumidification obviously makes the sterilization process complicated, it is not convenient to use; BDS-5000 is relatively relatively relatively relatively loose It is greatly adapted to various environmental conditions to ensure efficient use in all kinds of places and equipment.
Second, gas production
By specially designed "flash" technique, the BDS-5000 will generate 30-35% analysis of the purely grade hydrogen peroxide solution into a gaseous molecular state, which is a pampus (2.97 x 10e-12), The pure gas state, excellent diffusion; other equipment cannot be completely gaseous in this regard, containing a large amount of impurities, so the filter must be passed during discharge; when the resulting is not a pure gas, it (dry fog Or wet fog), both of them have essentially different. Of course, because there is a large defect in the atomization system, it is not accepted for most units.
Third, sterilization
1, hydrogen peroxide liquid concentration:
Some other brand devices use a hydrogen peroxide solution with a concentration of about 60%, which is extremely dangerous, greatly increasing the difficulty of control, and there is a great explosion risk at the time of operation; and due to high concentration The hydrogen peroxide solution produces condensation, when cooling, the liquid drop is placed on the machine panel, causing corrosion of the equipment itself;
In addition, the high concentration of hydrogen peroxide stock solution is also very difficult, and the safety requirements in transport will greatly limit the customer's use;
2, sterilizer concentration:
Other brands of equipment, its sterilization process is very weak to concentration, because mostly use dry sterilization, in the case where humidity is low enough, the concentration of hydrogen peroxide in the environment will be quickly out of control, which is large Leakage risk, and corrosion of materials;
3, sterilization efficiency:
In sterilization, due to the low humidity, hydrogen peroxide can only be irregular random motion, the efficiency is extremely low; the BDS-5000 is revealed, and there is a certain humidity (water molecule) in the surface of the particles, its HPV molecule can be "targeted "The surface is covered on the surface of the particles, forming a" micro-condensed "formation of a layer of naked eye to quickly and efficiently sterilizing, and its efficiency is significantly improved.
4, single cycle - double cycle
Other brands of equipment use single-cycle models, and the intentions are as follows:
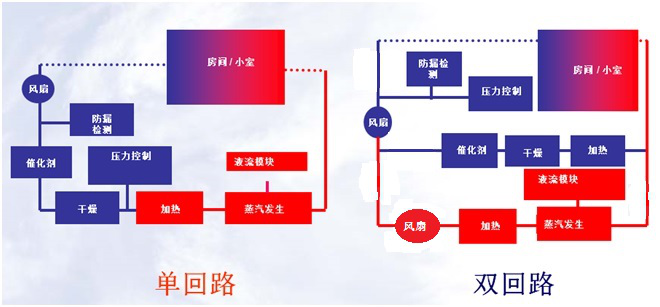
As shown in the figure, since the loop of the single loop will continue to decompose hydrogen peroxide, this exists two problems:
1) Consume more hydrogen peroxide and increase the cost of consumables;
2) When it is prolonging sterilization
safety: safety
From the perspective of EHS, other brands of equipment, without decomposition devices, must rely on the ventilation system of the room to assist; our company's equipment, such as the BDS-5000 is a decomposition unit with HPV, or through external type Ventilation degradation equipment decomposes HPV;
Material compatibility:
Although other brands of equipment claim that material is compatible, there is a large risk, such as due to non-pure gaseous, has a large humidity, so that electronic equipment is easy to produce short circuits, fires. Confused our company's equipment, the HPV steam sprayed, the naked eyes did not see, and the compatibility of most items, including sensitive electronic equipment;
Verified:
Our company's HPV technology has strong authentication, its HPV process comes with high concentration and low concentration HPV sensor, full monitoring, controllable, complies with GMP / GLP specification requirements, this other brand equipment is If you can't do it, you need to monitor other optional handheld auxiliary equipment, no online concentration monitoring, so you can't verify;
Actual Cases - A customer uses our three BDS-5000 hydrogen peroxide steam sterilifiers to sterilize the B-level area of the factory, and our company is as follows:
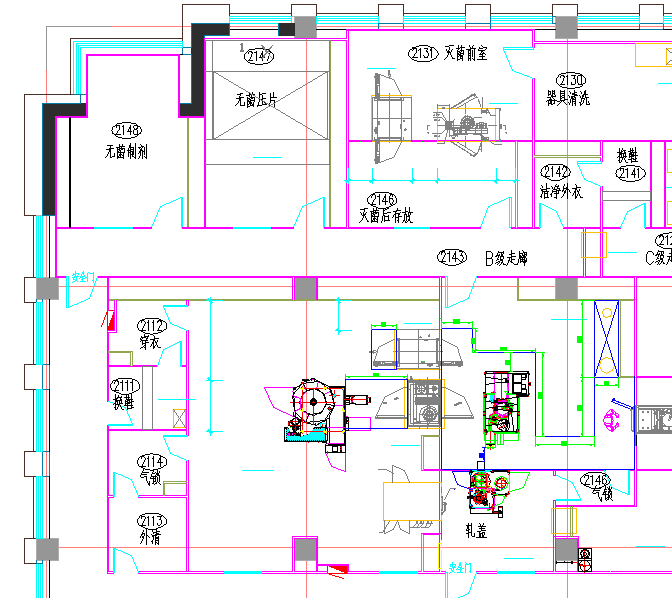
As shown in the figure, it is divided into three regions to be sterilized, and 1 BDS-5000 is used for sterilization:
1. Regions 2130 to 2148, about 117 square meters:
The steam generator is placed in the corridor;
The entire sterilization is about 4 hours
2, filling room: about 71 square meters
The steam generator is placed in the filling room, and the entire sterilization is about 2.5 hours.
3, rolling room: about 54 square meters
The steam generator is placed in the filling room, and the entire sterilization is about 2 hours.
The three regions were sterilized using 1 machine, and the total sterilization time required for approximately 9 hours, which is a night; if three machines are used to sterilize three regions, total sterilization Time is no more than 5 hours. It can be placed in the next time to open the sterilization, and the machine will be charged and produced on the next morning.

Scan it