
Time:2018-03-16| Author:admin
hydrogen peroxide peroxideWhy is it disinfected and bactericidal after high temperature?
The student of hydrogen peroxide is hydrogen peroxide, and a hydrogenant molecule is made of 2 hydrogen atoms and 2 oxygen atoms, and the chemical formula is H2O2. It can be seen from the chemical formula that a hydrogen molecule is more than one oxygen atom more than a water molecule. Hybride is a liquid of colorless and irritating odor. Medical usually 3% of the hydrogen peroxide is disinfected by wounds or otitis media.
Why is the hydrogen peroxide with disinfection and sterilization? This is because the peroxide is unstable, it is easy to happen the following reactions:
H2O2 → H2O (O)
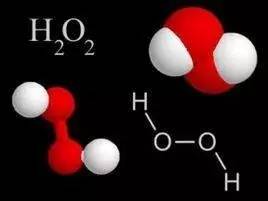
When it meets the wound, pus or dirt, immediately decompose the formation of oxygen. This oxygen atom that has not yet binds to oxygen molecules has strong oxidation capacity. When contacting bacteria, it can destroy bacterial bacteria and kill bacteria.
After killing bacteria, the remaining substance is no poison, no stimulating water. No secondary pollution is formed. Therefore, hydrogen peroxide is a disinfectant for wound disinfection. However, it is not possible to disinfect the wound in a concentration of hydrogen peroxide to prevent burning skin and affected area.
Hydrogen peroxide (H2O2) is a product that can be bought in pharmacies. If you purchased a 3% solution, 97% water and 3% hydrogen peroxide were contained in the bottle. Most people use it as preservatives. It is not a good preservative, but it is a good choice for cleaning the cutting and rubbing, and the resulting foam looks cool.
Hydrogen peroxide will be foam because blood and cells contain enzymes called catalase. Since the cut or wound has both blood flow, there will be damaged cells, so a large amount of catalase is concentrated around the wound.
When the hydrogen peroxide is in contact with the hydrogen peroxide, it converts hydrogen peroxide (H2O2) into water (H2O) and oxygen (O2).
The efficiency of the above reaction is extremely high - 200,000 reactions per second. The bubbles seen in the foam are pure oxygen bubbles produced by catalase. Try to add a little hydrogen peroxide on the cut potato or observe the same phenomenon, the same - the same - damaged hydrogen peroxide in the damaged potato cells reacts with the hydrogen peroxide.
Hydrogen peroxide does not produce bubbles on the bottle or in the skin because there is no catalase to promote the occurrence of the reaction. Hydrogen peroxide is very stable at room temperature.
Shenzhen Fukang Technology Co., Ltd.With a number of patented technologies, the high-temperature hydrogen peroxide steam produced by hydrogen peroxide is evaporated by the generator until the hydrogen peroxide vapor saturation in the space is reached, and the high temperature saturated hydrogen peroxide vapor is contacted. The surface of the sterilized article will reach the micro-condensation state, and the microbial enveloping layer is formed on the surface of various microorganisms, and the hydrogen peroxide molecules will react with light or metal ions, release strong oxidation resistance, when contacting the cells Destroying the cell membrane and DNA, free radicals inactive, killing microorganisms in a very short period of time, achieving disinfective effect, free radicals can reach 6-log killing effects on microorganisms.

Scan it