
Time:2018-03-16| Author:admin
Vaporization steamingSterilization of hydrogen peroxideAt present, most users are called "VHP\\ But in fact, VHP is a registered trademark of this company's sterilization product. At regulations, USP is called "VPHP, VAPOR PHASE HYDROGEN PEROXIDE". In any case, "Vapor" has been defined as the state of this sterilization medium, then what state is VAPOR? Is gas (GAS) or steam (Steam)? What is the factor affecting sterilization effect during sterilization? These issues will be described herein.
What is Vapor?
We all know that the substances have three states, solid (liquid), gas (GAS). Vapor, scientific definitions is that the gaseous state is in a state where the temperature is lower than its critical point, while the gaseous and liquid state is present. When the temperature is below the dew point temperature, the gaseous is condensed into fog, and even the droplets can be condensed on the low temperature surface.
Regardless of the vaporized hydrogen peroxide generator, the resulting hydrogen peroxide Vapor is a mixture of H2O2 and H2O in both gaseous and liquid states [1]. Some people may think that hydrogen peroxide produced by the generator is a gas (GAS) that can be seen in the naked eye is not a visible steam (Steam). However, in fact, the boiling point of 30% to 35% of the oxygen-vaporization solution is 108 ° C, that is, when the temperature is temperature to reach a temperature of 108 ° C or more, it can be maintained in the gas state. This is impossible under the environmental conditions of practical applications. The picture below shows the boiling point of the concentration of hydrogen peroxide solutions in different (mass percent).
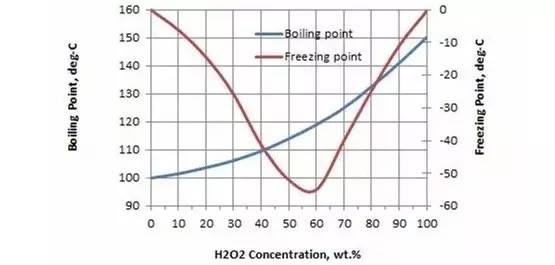
Another vaporizationSterilization of hydrogen peroxideThis way is a "mysterious" reason, which is generated in a state of unstable and is always changing.
From a physical point of view, the hydrogen peroxide solution is transmitted to the H2O2 and H2O mixed steam (gas) generated by vaporization to the target environment in which to sterilize it, and after mixing with the air in the target environment, strive to be in new Renained balance under environmental conditions. This balance is achieved by the mutual transition of the gas state and the liquid state under conditions of temperature and humidity.
From a chemical perspective, the hydrogen peroxide vaporification is more likely to decompose, and both of H2O and oxygen are obtained.
From the microbial reaction, the presence of microorganism accelerates the decomposition of hydrogen peroxide, and the microorganism is killed under its oxidative effect.
This is the unstable state, so that we have little way to know the concentration under each location under the existing technical conditions. But fortunately, hydrogen peroxide can have the ability to kill the microorganisms in gas and liquid. And there is theoretical basis for support in Vapor state, the liquid-state H2O2 relative gaseous H2O2 killing effect is faster.
The concentration under the H2O2 liquid conditions will always be higher, which is determined by the surface vapor pressure of the H2O2 solution. Regardless of whether we are accepting, the Vapor of H2O2 and H2O in the sterilization process will exist in the sterilization environment, just how much problems. The H2O2 is condensed first than H2O, which allows the hydrogen peroxide concentration of the object surface to be higher than the solution concentration of 30% hydrogen peroxide before the evaporation [2].
What are the factors of sterilization effect?
Under the premise of understanding what the VAPOR status and the mechanism of condensation will occur during its sterilization process, the factors affecting the sterilization effect will naturally be derived.
First, from the condition of generating H2O2 and H2O VAPOR, the temperature of the VAPOR produced, the concentration of the hydrogen peroxide solution determines the state of the VAPOR. Among them, the evaporation rate of hydrogen peroxide will also affect the sterilization effect.
Second, the modification of the VAPOR state of H2O2 and H2O is determined by transmitting a sterilized gas into a target environment (isolator, room). For example, using pipeline transmission, if no heat is carried out, the background ambient temperature is relatively low, then sterilization steam is condensed inside the pipe. H2O2 contained in Vapor delivered to the target environment will be relatively lowered.
In addition, the airflow speed and airflow forms are transmitted to the environment.Sterilization of hydrogen peroxideThe unevenness of the gas distribution.
Third, the temperature of the target environment is too high or the temperature is uneven, which causes the sterilized gas to condense unevenness in the surface. General temperatures /-2.5 C can be accepted. It should be noted that the high temperature surfaces in those environments, such as CIP / SIP functions such as lyophilization machines, filling machines, etc., which can cause high temperatures of local surfaces, need to be cooled (generally less than 40 ° C) to vaporizeSterilization of hydrogen peroxide[3]. In addition, the high temperature will change the state in the environment due to heat radiation, so that the local peroxide vaporization gas cannot reach these parts.
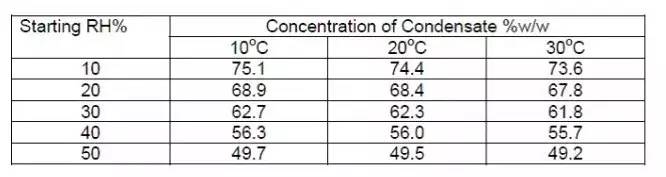
Fourth, the initial relative humidity of the target environment is too high, and the proportion of H2O containing H2O is increased after mixing with H2O2 and H2O sterilization gas, so that the condensation produced during the sterilization process may excessively, and the condensed hydrogen peroxide liquid concentration Relatively low. The following figure shows the concentration of hydrogen peroxide concentrations in different temperatures and initial humidity conditions [4]. The uniformity of humidity will be affected by temperature uniformity, and the surface of the temperature is relatively localized is low.
Fifth, vaporized hydrogen peroxide concentration. During the sterilization process, hydrogen peroxide itself is constantly being produced and is constantly decomposed. Maintaining hydrogen peroxide at a certain level is the approach of most generator manufacturers, and some are based on concentrations, and some are considered in condensation. No matter which detection method exists, "how to choose a representative detection site" problem. This is indeed the difficulty of this technology. Due to the limitations of detection techniques, most of the electrochemical hydrogen peroxide concentration sensors are currently used as detection means.
It should be noted that the sensor is only the concentration of hydrogen peroxide in the space in the space, but the surface condensation concentration is unknown, and the truly active is the unknown hydrogen peroxide. The limitations of hydrogen peroxide concentration in the detection space can be seen from the following two curves. (A) Due to the large coagulation amount, a portion of the hydrogen peroxide is condensed to the surface, and the amount of hydrogen peroxide present in the space is reduced, which is why continuous injection of hydrogen peroxide, but the amount detected is increasing The less. (B) In the figure, the coagulation amount is less, so the hydrogen peroxide concentration can always be maintained at a higher level. As you can see, the concentration in the space cannot directly explain the quality of the sterilization effect. However, as a parameter of a sterilization cycle, it is necessary to detect its concentration. Problems in sterilization cycles can be analyzed by concentration data analysis.
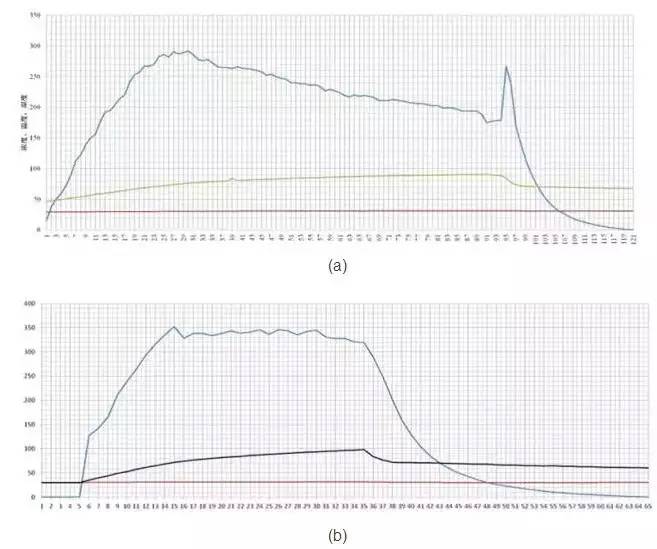
Sixth, the amount of hydrogen peroxide is more reliable in the sterilization process and more controllable parameters. Therefore, the total amount of hydrogen peroxide is a key parameter for sterilization. Different temperatures were compared to different temperature and initial environment humidity conditions in Document [5], the amount of microbial killing was contracted. Conclusion, regardless of the relative humidity of the environment, how the temperature changes, it ultimately determines whether the parameters that can reach the microbial killing is the amount of hydrogen peroxide.
In short overall
1. Vaporization hydrogen peroxide is a mixture of hydrogen peroxide and water in liquid and gaseous, and is unavoidable during sterilization, but excess condensation is to be avoided.
2. The concentration does not directly affect the parameters of the sterilization effect.
3. Sterilization effect is the concentration of hydrogen peroxide solution, vaporization, hydrogen peroxide solution, target ambient temperature and humidity conditions, load conditions, material [6], etc.

Scan it